Human Growth Hormone (hGH)- Purification and Uses
This blog post gives an overview of the uses and features of hGH, as well as its optimized soluble purification using the CL7/Im7 system.
Human growth hormone (hGH), also known as somatropin, is a hormone secreted by the pituitary gland. Recombinant protein expression methods have allowed the 22 kDa protein to be produced in a laboratory setting. hGH is relevant in a wide range of biological functions and is used in the treatment of diseases such as HIV wasting syndrome, hypopituitary dwarfism, and genetic disorders. Attempts have been made to utilize hGH for performance-boosting purposes, but self-experimentation is dangerous and does not always result in the desired effects.
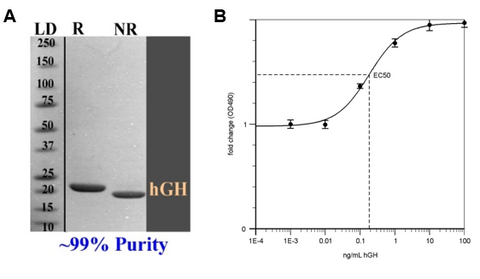
hGH for clinical research comes in lyophilized and liquid forms. It is commonly available in both naturally isolated and recombinant forms. Markers of high quality hGH are ultra-high purity (>98%) and a low EC50. EC50 denotes the concentration of the protein/substance to produce 50% of the maximum effect. TriAltus’ hGH is >99% pure and has an EC50 of ~0.2 ng/mL.
hGH-CL7 Purification
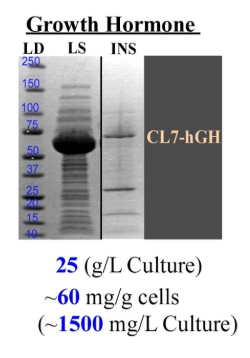
Purification of hGH from E. coli has traditionally been difficult because the protein tends to be insoluble due to inclusion body aggregation (Figure 1). Inclusion bodies require denaturing and refolding steps in the purification process which can impact the structural integrity of the final product.
Figure 1. Amount of insoluble hGH (INS) compared to amount of protein in starting lysate (LYS). 25 g E. coli/L culture yielded ~60 mg/g cells (~1500 mg/L culture).
When expressed without a tag in E. coli, hGH is insoluble due to misfolding, likely from an incorrectly formed internal disulfide bond.. With a CL7 expression tag, among others, however, the protein is recoverable in soluble form. CL7 enhances expression and helps with stability, solubility and folding of proteins expressed in E. coli. This avoids the need for additional refolding and chromatography steps.
Purification using the CL7/Im7 system not only solubilizes hGH, it results in high yield, purity, and activity protein. From 1 L of cell culture, 1500 mg of hGH was recovered (Figure 1). In just one chromatography step, near-perfect purity was achieved of >99%. SDS-PAGE analysis of the protein in both reduced and non-reduced conditions indicate proper disulfide bond formation. (Figure 2A). TriAltus hGH exhibited in vivo biological activity identical to that of other commercial/clinical samples, with an EC50 of ~0.2 ng/mL measured by a cell proliferation assay using 32D-rGHR cells (Figure 2B).
Figure 2. A) >99% purity hGH protein in reduced (R) and non-reduced (NR) forms after one chromatography step. B) EC50 activity graph for TriAltus-purified hGH.