The SUMO protease releases target proteins from TriAltus’s Im7 resin. This protease recognizes SUMO tertiary structure – not a specific amino acid sequence – to cleave and release the target protein from the Im7-bound CL7 tag.
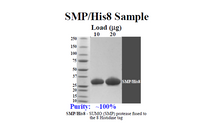
PURITY | ~100% (SDS gel analysis)
ENZYME SOURCE | Yeast protein overexpressed in E. coli with histidine (His) tag
CLEAVAGE SITE | SUMOP cleaves after the C-terminal glycine at the end of the SUMO sequence. If the target protein is fused downstream of the SUMO C-terminus, SUMOP cleavage renders the native protein sequence without any additional amino acids.
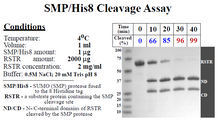
CLEAVAGE EFFICIENCY | 1 ug protease will digest 2 mg protein with 99% efficiency at 4°C in 40 minutes.
STORAGE/SHIPPING CONCENTRATION | ~7.5 mg/mL
SHIPPING CONDITIONS | We ship SUMO protease on dry ice in buffer (15 mM Tris (pH 8.0), 0.2 M NaCl, 50% glycerol).
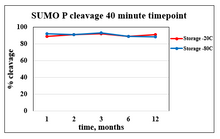
RECOMMENDED STORAGE CONDITIONS | -80°C long term or -20°C for 2-3 months in shipping buffer
EXPIRATION | 6 months from receipt when stored as directed
SKU |
1 mg: 30-1130
5 mg: 30-1135
10 mg: 30-1140
50 mg: 30-1146
100 mg: 30-1150
Additional processing information: SUMOP releases CL7-tagged proteins from the Im7 resin-bound CL7 tag. The final protease concentration in the eluted sample comprises ~1.5-3% of total impurities. If needed, SUMOP can be purified from the sample with a His-trap column or via size exclusion chromatography.
Use of TriAltus products falls under our limited license agreement and is for non-profit research use only. For commercial use, please contact us for licensing.